Medical Industry
Medical Plastic Injection Molding
- 100,000-class (ISO 8) dust-free workshop, certified for PVC medical molding
- 316L medical-grade stainless steel, resistant to chlorine-based disinfectants
- Complies with FDA and EU MDR standards, real-time environment monitoring
TDL: A Leading Medical Plastic Injection Molding Company in China!
TDL specializes in high-quality plastic injection molding for medical and laboratory applications. With an ISO 8 clean room and full sterile management processes, TDL has become a premium supplier of core medical consumables such as tips and luer tubes.
Medeical Device Injection Molding Services

Mold Design
We provide customized mold design solutions tailored to various medical products, such as syringe components, implants, and diagnostic equipment parts. Utilizing advanced 3D modeling and simulation tools, including Moldflow analysis, we optimize gating systems, cooling channels, and mold structures to ensure efficient production and consistent quality.

Mold Manufacturing
TDL specializes in producing medical component molds with strict tolerances, supporting injection molding processes. Our injection mold manufacturing employs high-precision machining technologies, including precision milling, turning, wire EDM, and electrical discharge machining, to ensure exceptional dimensional accuracy and surface quality of the molds.

Injection Molding
We utilize biocompatible materials such as PC, PEEK, and LCP to produce medical-grade plastic components. Our cleanroom manufacturing environment ensures contamination-free production. Our injection-molded medical parts are widely used in applications such as medical device housings, diagnostic equipment components, and disposable medical products.
Applications
Common Medical Industry Applications
Explore the world of healing with TDL: Your trusted partner for medical device manufacturing.

Medical Disposable

Life Science Product

Beauty Equipment

Medical Instrument

Medical Devices
Medical Injection Molding Materials
We use biocompatible, durable, and chemically resistant medical-grade materials that comply with global certifications and strict quality standards, including FDA and ISO 13485. We also customize material selection to meet your specific requirements.

Polycarbonate (PC)
Excellent impact resistance, clarity, and strength, ideal for medical devices requiring high durability and transparency.
Applications: Syringes, dialysis equipment, drug delivery systems, monitoring instruments, surgical tools, etc.

Polypropylene (PP)
Lightweight, chemically resistant, and cost-effective material, especially suitable for single-use medical products.
Applications: Disposable syringes, drug containers, infusion sets, surgical tools, instrument trays, etc.

Polyurethane (PU)
Highly elastic, flexible, and chemically resistant. Widely used in medical devices that require high durability and flexibility.
Applications: Catheters, vascular implants, balloons, wound dressings, etc.

Polytetrafluoroethylene
Superior chemical resistance, low friction properties, and high-temperature resistance. Its biocompatibility makes it suitable for long-term implantable devices.
Applications: Surgical instruments, catheters, implants, seals, etc.

Polyetheretherketone (PEEK)
High-performance engineering plastic with excellent high-temperature resistance, chemical stability, and mechanical strength. Well-suited for medical devices that must bear heavy loads or perform in demanding environments.
Applications: Orthopedic implants, dental implants, surgical instruments, artificial joints, etc.
Silicone
Excellent biocompatibility, high-temperature resistance, and flexibility. Ideal for medical products that require softness, flexibility, and long-term body contact.
Applications: Respirators, catheters, chest drainage tubes, prosthetics, etc.

Polyethylene (PE)
High biocompatibility, chemical stability, and wear resistance. Ideal for many long-term medical devices. Its flexibility and processability also make it suitable for a wide variety of medical applications.
Applications: Implants, catheters, infusion tubing, dialysis tubing, etc.

Polyvinyl Chloride (PVC)
Economical, easy-to-process plastic material with good chemical resistance, wear resistance, and UV stability. Widely used in a variety of medical products.
Applications: Infusion tubing, blood bags, urinary catheters, surgical instruments, etc.
Advantages
Why ChooseTDL for Medical Device Injection Molding?
Certified
TDL is a ISO 13458 certified mold manufacturer with a strong track record of quality and performance.
Medical Grade Plastic Materials
TDL uses medical grade plastic materials for molds that are used in medical applications, ensuring that all products meet the highest standards of safety and effectiveness.
Provide CPK Test
TDL provides CPK (Process Capability Index) tests to ensure that the injection molding process is capable of producing products that meet the customer's specifications.
Cleanroom Production
TDL operates a 100,000-class (ISO 8) dust-free workshop, certified for PVC medical molding. Combined with 316L stainless steel tooling, resistant to chlorine-based disinfectants, this ensures contamination-free production.
Mold Validation
Comprehensive mold acceptance tests (FAT/SAT) are carried out before delivery, confirming compliance with design and performance requirements.
Good After-sales Service
TDL is responsible for quality of each mold, will sign mold quality warranty with customers for every tools.
How Do We Ensure the Quality and Compliance?
As a medical device injection molding manufacturer, we deeply understand the critical importance of ensuring product quality and compliance. This is not just about the product itself, but also about patient safety.
Selection of Compliant Medical-Grade Materials
FDA-Approved Materials: We meticulously select FDA-approved medical-grade plastics to ensure they meet biocompatibility, non-toxicity, and corrosion resistance requirements. These materials are suitable for manufacturing a wide range of medical device components.
Material Traceability: We maintain detailed traceability records for each batch of raw materials, ensuring that the source and production process of every injection-molded part can be traced. This guarantees the safety and compliance of the products.


Sterilization and Packaging Compliance with Medical Standards
Sterilization Process Validation: We rigorously control the sterilization process for each injection-molded component, using approved sterilization methods such as autoclaving (steam sterilization) and gamma radiation sterilization. This ensures product sterility, meeting the stringent requirements for medical device usage.
Cleanroom Production Environment: To prevent contamination, our medical injection molding production lines are typically located in cleanroom environments. This ensures that the products remain uncontaminated during production, in compliance with ISO 14644-1 cleanroom standards.
Rigorous Quality Control and Testing
We implement stringent quality control at every stage of the manufacturing process, encompassing comprehensive inspection of molds, injection molding parameters, and finished goods. Statistical Process Control (SPC) is employed to monitor critical process parameters, ensuring process consistency and stability.
Sterility Testing: For medical devices requiring sterility, we conduct rigorous sterility testing to ensure the achievement of the predetermined Sterility Assurance Level (SAL). Testing methods comply with relevant regulations and standards.
Biocompatibility Testing: Comprehensive biocompatibility testing, including in vitro and in vivo studies, is performed on the final product to verify biocompatibility and ensure no adverse reactions in patients.
Other Testing: Depending on specific product requirements, we perform other necessary tests, such as dimensional measurements, leak testing, etc., to ensure the product meets design specifications and performance requirements.


Strict Adherence to Industry Standards and Regulations
ISO 13485 Certification: As a specialized medical injection molding company, we are ISO 13485 certified and have established a rigorous Quality Management System (QMS) to ensure our production processes meet international medical device standards.
FDA Compliance: Our manufacturing processes strictly adhere to the FDA’s Quality System Regulations (QSR), ensuring that all injection-molded medical components comply with FDA safety and efficacy requirements for medical devices.
CE Marking and MDR Compliance: In the European market, we ensure our products comply with the EU Medical Device Regulation (MDR) and obtain CE marking certification to meet market entry requirements.
The Benefits of Injection Molding Process for Medical Devices
High Precision and Consistency
Injection molding, through precision molds and automated production processes, ensures that every product in mass production meets strict tolerance standards.
Cost-Effectiveness
Once the mold is developed, injection molding enables fast, high-volume production, significantly reducing unit costs, making it ideal for disposable consumables such as test tubes and IV components.
Material Versatility
Injection molding supports a wide range of medical-grade plastic materials, which can be selected based on specific product requirements, such as biocompatibility, chemical resistance, transparency, or flexibility.
Compliance and Traceability
The production process is easy to control and complies with regulatory standards such as FDA and CE. Molds can also integrate serial numbers or barcodes, enabling full lifecycle traceability for each product.
Medical Plastic Injection Molding Team
Meet Our Expert Team: The Driving Force Behind TDL Mold’s Innovative Solutions.
Our Equipment

CMM
Inspection

High Speed CNC Machines

Double-Head EDM Machines

EDM
Workshop

CNC machining Workshop

Plastic Injection Molding Room

Mirror EDM Machining

Mould Spotting Machine
Choose us for medical injection molding and get up to 100% mould cost refund!
FAQ's

Plastic molds are widely used in the manufacturing of various medical devices due to their versatility, precision, and cost-effectiveness. Some examples of medical devices that are commonly made using plastic molds include:
- Syringes: Plastic molding is commonly used to create the components of syringes. Injection molding, a precise and efficient technique, is often employed to produce the barrel, plunger, and cap of syringes. The high precision of plastic molds ensures consistent dimensions and smooth surfaces, which are crucial for accurate dosing and patient comfort.
- IV Catheters: Intravenous catheters are essential for delivering fluids and medications directly into the bloodstream. The manufacturing of these catheters involves plastic molding to create the catheter hub, wings, and needle guard. Using molds enables the production of components with consistent dimensions, reducing the risk of discomfort and complications during insertion.
- Medical Tubing: Various types of medical tubing, such as oxygen tubes, drainage tubes, and catheter tubing, are manufactured using plastic molds. These molds allow for the creation of tubing with specific diameters, wall thicknesses, and surface finishes, ensuring compatibility with medical equipment and patient needs.
- Respiratory Masks: Respiratory masks used in medical settings, including oxygen masks and nebulizer masks, often have plastic components produced through molding. The molding process allows for the precise shaping of mask components, ensuring a secure fit and effective delivery of respiratory therapies.
- Blood Collection Tubes: Plastic molding is utilized to create blood collection tubes used for various diagnostic tests. The consistent quality of molded tubes is crucial to maintain the integrity of blood samples and accurate test results.
- Implantable Devices: While not all implantable medical devices are made using plastic molds, certain components of implantable devices, like surgical mesh used in hernia repairs, can be manufactured using molding techniques. The use of medical-grade plastics and precise molding processes ensures biocompatibility and structural integrity.
- Diagnostic Devices: Diagnostic devices often include plastic components produced through molding. These components can include housings for diagnostic equipment, test cartridge holders, and sample wells. The high level of precision achievable with plastic molds is essential for reliable test results.
- Lab Equipment: Plastic molding is employed in the production of laboratory equipment such as petri dishes, microplates, and sample vials. The consistency and quality of molded lab equipment are vital for accurate research and experimentation.
- Dental Devices: In the field of dentistry, plastic molding techniques are used to create a range of devices, from dentures and orthodontic aligners to mouthguards. The ability to customize the shape, size, and fit of these devices through molding enhances patient comfort and treatment outcomes.
- Disposable Medical Devices: Many disposable medical devices, such as gloves, masks, and wound dressings, are made using plastic molding processes. The efficiency of molding allows for large-scale production of these essential items while maintaining quality and cost-effectiveness.
- Prosthetics and Orthotics: Some components of prosthetics and orthotics, particularly those that require lightweight and durable materials, can be manufactured using plastic molding. This enables the creation of functional and comfortable devices for patients with limb differences or musculoskeletal conditions.
- Diagnostic Kits: Plastic molding is utilized to create various components of diagnostic test kits. These include reagent holders, sample wells, and housing for test strips. The precision of molding ensures accurate results and ease of use for healthcare professionals.
- Drug Delivery Devices: Devices used for drug delivery, such as inhalers, insulin pens, and auto-injectors, often involve plastic molded parts. These devices require precise engineering to ensure accurate dosing and patient safety.
- Surgical Instruments: While many surgical instruments are made from metals, certain plastic components in surgical tools can be produced using molding techniques. These components might include handles or grips for improved ergonomics and hygiene.
In the medical field, the choice of plastic material, the molding technique, and adherence to regulatory standards (such as ISO 13485 for medical devices) are critical factors in ensuring the safety, efficacy, and quality of the devices produced. Plastic molding offers numerous advantages, including scalability, repeatability, and the ability to create intricate and customized designs, making it a cornerstone of medical device manufacturing.
Source: https://youtu.be/vN_Bv7079jg
Plastic molds are essential tools in the production of various medical equipment. They enable the precise and efficient manufacturing of intricate components used in medical devices. The process involves several steps:
- Design and Engineering: The design phase involves creating detailed CAD models of the medical equipment components. Engineers work closely with medical professionals to ensure that the design meets the functional requirements of the device and adheres to relevant standards and regulations. Considerations include not only the physical shape but also factors like ease of use, ergonomics, and compatibility with other medical equipment.
- Material Selection: Medical-grade plastics are selected based on their biocompatibility, sterilization capabilities, and mechanical properties. Plastics used in medical device manufacturing must meet stringent quality and safety standards. The choice of material depends on factors such as the device's intended use, its exposure to chemicals or bodily fluids, and whether it needs to be transparent or opaque.
- Mold Fabrication: The fabrication of molds is a precise and skilled process. Molds can be manufactured using materials like steel or aluminum. The mold design includes cavities and cores that define the shape of the final component. Complex molds might include intricate features, such as undercuts, threads, or microstructures. Highly specialized machinery and techniques, like CNC (Computer Numerical Control) machining or electrical discharge machining, are used to create the mold with high accuracy.
- Molding Process:
- Clamping: The mold is securely clamped shut using hydraulic or mechanical clamps to prevent any leakage of plastic material during the injection process.
- Injection: Plastic pellets or granules are fed into an injection molding machine's hopper. These pellets are then heated and melted in a barrel before being injected under high pressure into the mold cavity. Injection pressure and temperature are carefully controlled to ensure proper filling of the mold.
- Cooling: Once the melted plastic has taken the shape of the mold cavities, it is allowed to cool and solidify. Cooling time is crucial, as it affects the material's properties and dimensional accuracy.
- Ejection: After cooling, the mold is opened, and the solidified parts are ejected from the mold using ejector pins. Ejection mechanisms are designed to avoid damaging the parts during removal.
- Cycle Repeats: The entire process of clamping, injection, cooling, and ejection constitutes one molding cycle. This cycle is repeated for each set of components to be manufactured.
- Quality Control and Testing: Quality control is a critical step in medical device manufacturing. Components are thoroughly inspected for dimensional accuracy, surface finish, and other specifications. Some components might undergo testing for material properties, biocompatibility, chemical resistance, and mechanical performance. These tests ensure that the components meet the necessary standards and will perform as expected in medical applications.
- Post-Processing: Depending on the requirements of the medical equipment, post-processing steps might include:
- Trimming: Excess plastic or flash is removed from the molded components.
- Assembly: If the device consists of multiple components, they might be assembled using techniques like ultrasonic welding, adhesive bonding, or snap fits.
- Surface Treatment: Components might undergo treatments like polishing, surface texturing, or printing of labels or identification marks.
- Sterilization: Many medical devices require sterilization to ensure they are free from pathogens. Sterilization methods can include gamma irradiation, ethylene oxide gas, autoclaving, or chemical sterilization.
- Packaging and Distribution: Proper packaging is crucial to maintaining the sterility and integrity of the components until they are used. Packaging materials must be designed to withstand sterilization processes and ensure that the components are protected during transportation and storage.
- Regulatory Compliance: Throughout the entire process, adherence to regulatory standards specific to medical devices is paramount. Depending on the region, these standards might include ISO 13485 for quality management systems, FDA regulations (in the United States), and CE marking (in the European Union). Compliance ensures that the manufactured medical devices are safe, effective, and of consistent quality.
Plastic molding plays a pivotal role in the efficient and precise manufacturing of medical equipment components. It combines engineering expertise, material science, and meticulous quality control to produce devices that meet the highest standards of performance and safety in healthcare settings.

Image courtesy: https://www.starrapid.com/blog/the-ten-most-popular-plastic-injection-molding-materials/
The choice of materials for medical plastic molds is crucial, as they must possess specific properties to withstand the demands of the molding process and produce high-quality medical device components. Common materials used for medical plastic molds include:
- Polypropylene (PP):
- Properties: Polypropylene is a thermoplastic known for its exceptional chemical resistance, high melting point, and low density. It exhibits good impact strength and can be molded with ease.
- Advantages: PP's resistance to chemicals, heat, and impact makes it a valuable choice for medical applications. It is commonly used for devices requiring a balance between mechanical properties and affordability.
- Applications: In medical plastic molds, PP finds its place in producing items like syringes, medical bottles, laboratory containers, and packaging due to its durability, chemical resistance, and cost-effectiveness.
- Polyethylene (PE):
- Properties: Polyethylene, available in different densities (LDPE, HDPE), is a versatile thermoplastic with excellent chemical resistance, electrical insulation, and low friction properties.
- Advantages: PE's flexibility and biocompatibility make it suitable for medical applications. It's used in devices requiring a wide range of mechanical properties and can be molded or extruded with relative ease.
- Applications: Medical bags, tubing, surgical drapes, joint replacements, and even implants benefit from PE's versatility and biocompatibility.
- Polystyrene (PS):
- Properties: Polystyrene is a rigid, clear thermoplastic known for its transparency, hardness, and ease of molding. It can be either crystal clear or modified for impact resistance.
- Advantages: PS's optical clarity and rigidity make it ideal for applications requiring transparency, such as laboratory equipment, petri dishes, and diagnostic components.
- Applications: Molds made from PS are commonly used for items like microscope slides, culture dishes, and clear diagnostic components that require visual inspection.
- Polycarbonate (PC):
- Properties: Polycarbonate is a strong, transparent thermoplastic known for its exceptional impact resistance, optical clarity, and high heat resistance.
- Advantages: PC's impact resistance and transparency make it a top choice for medical devices requiring both durability and visibility.
- Applications: Surgical instruments, connectors, transparent housings for medical electronics, and components that require impact resistance benefit from PC's properties.
- Polyvinyl Chloride (PVC):
- Properties: PVC is a versatile thermoplastic known for its flexibility, durability, and chemical resistance. However, it may require plasticizers to achieve certain properties.
- Advantages: PVC's flexibility makes it suitable for applications requiring pliability, such as medical tubing and bags.
- Applications: Despite its wide use in medical tubing, blood bags, and IV bags, PVC is seeing a decline in some areas due to concerns about plasticizer leaching and efforts to find safer alternatives.
- Polyetheretherketone (PEEK):
- Properties: PEEK is a high-performance thermoplastic known for its excellent mechanical properties, high heat resistance, and biocompatibility.
- Advantages: PEEK's strength, stability, and biocompatibility make it ideal for implantable medical devices where exceptional performance and biocompatibility are paramount.
- Applications: PEEK is used in medical molds for producing orthopedic implants, spinal fusion cages, and other components requiring a combination of mechanical strength and biocompatibility.
- Polyethylene Terephthalate (PET):
- Properties: PET is a strong, lightweight thermoplastic known for its excellent barrier properties, dimensional stability, and clarity.
- Advantages: PET's strength and barrier properties make it suitable for preserving sterility in medical packaging and disposable medical devices.
- Applications: PET is commonly used in molds for producing medical packaging, containers, and disposable devices where maintaining the integrity of the contents is crucial.
- Polyurethane (PU):
- Properties: Polyurethane is a versatile thermoplastic known for its flexibility, durability, and excellent abrasion resistance. It can be formulated to exhibit a wide range of properties.
- Advantages: PU's flexibility and durability are valued for medical applications that require comfort, resilience, and resistance to wear.
- Applications: PU is used in molds for producing catheters, medical tubing, wound dressings, medical gowns, and other devices requiring flexibility, durability, and comfort.
Certainly, here's additional information about other commonly used materials in medical plastic molds:
9.Acrylonitrile Butadiene Styrene (ABS):
- Properties: ABS is a thermoplastic known for its toughness, impact resistance, and ease of processing. It can be modified for different properties.
- Advantages: ABS's impact resistance and versatility make it suitable for medical device components that require both strength and ease of molding.
- Applications: ABS molds are used for producing a variety of medical equipment parts, housings, and structural components.
10.Polyoxymethylene (POM or Acetal):
- Properties: POM is a thermoplastic known for its high stiffness, low friction, and excellent dimensional stability.
- Advantages: POM's low friction and mechanical properties make it suitable for producing precise moving parts in medical devices.
- Applications: POM molds are used for components like gears, bearings, and other moving parts in medical devices.
11.Polymethyl Methacrylate (PMMA or Acrylic):
- Properties: PMMA is a transparent thermoplastic with good optical clarity, high UV resistance, and scratch resistance.
- Advantages: PMMA's transparency and optical properties make it suitable for applications requiring clear visibility and protection against UV radiation.
- Applications: PMMA molds are used for items like optical lenses, protective visors, and medical device covers requiring optical clarity.
12.Liquid Silicone Rubber (LSR):
- Properties: LSR is a silicone elastomer that can be molded to create flexible, durable, and biocompatible parts.
- Advantages: LSR's flexibility, biocompatibility, and resistance to heat make it suitable for producing medical components that require softness and comfort.
- Applications: LSR molds are used for medical devices like silicone seals, gaskets, soft-touch grips, and wearable medical technology.
13.Thermoplastic Elastomers (TPE):
- Properties: TPEs are flexible materials with rubber-like properties, combining the characteristics of both plastic and rubber.
- Advantages: TPEs provide flexibility, comfort, and resistance to chemicals, making them suitable for medical applications requiring a combination of properties.
- Applications: TPE molds are used for producing medical products like medical tubing, seals, gaskets, and grips requiring a balance of flexibility and durability.
14.Fluoropolymers (PTFE, FEP, PFA):
- Properties: Fluoropolymers offer excellent chemical resistance, low friction, and high-temperature stability.
- Advantages: Fluoropolymers' unique properties make them suitable for medical applications requiring resistance to harsh chemicals and extreme temperatures.
- Applications: Fluoropolymer molds are used for producing components like lab equipment, valves, and parts exposed to aggressive chemicals.
15.Polyethylene Naphthalate (PEN):
- Properties: PEN is a high-performance thermoplastic with excellent barrier properties, chemical resistance, and heat stability.
- Advantages: PEN's barrier properties and resistance to chemicals make it suitable for medical packaging and devices requiring excellent moisture and gas barrier.
- Applications: PEN molds are used for producing medical packaging, containers, and devices where preserving the sterility of the contents is vital.
In the realm of medical plastic molds, each material brings its unique set of properties and advantages to cater to specific device requirements. The selection process involves a meticulous evaluation of these properties, the intended application, regulatory compliance, and the desired performance of the final medical product. This comprehensive approach ensures that medical devices uphold the highest standards of safety, efficacy, and reliability.

Image courtesy: https://www.fictiv.com/articles/the-standards-for-plastic-injection-molding-in-the-medical-industry

Image courtesy: https://www.healthcarepackaging.com/markets/medical-devices/-packaginig/news/21415536/aptar-csp-technologies-aptar-csp-technologies-earns-iso-quality-certifications-for-medical-device-production
Regulations and standards play a significant role in shaping the design and manufacturing processes of medical plastic molds. These guidelines are in place to ensure the safety, quality, and performance of medical devices, including those produced using molds. Here's how regulations and standards impact the design of medical plastic molds:
- Material Selection: Regulatory bodies often specify the types of materials that are acceptable for use in medical devices. Medical-grade plastics and other materials must be chosen based on their biocompatibility, chemical resistance, and ability to withstand sterilization methods. The mold material must not introduce any contaminants that could affect the quality or safety of the final medical device.
- Biocompatibility: Medical devices come into contact with patients' bodies, so molds and the materials they are made from must meet biocompatibility standards. This ensures that components produced using the molds do not cause adverse reactions or harm to patients. The mold materials should not leach harmful substances into the plastic resin being molded.
- ISO 13485: ISO 13485 is an international standard that outlines the requirements for a quality management system specific to the medical device industry. Manufacturers of medical molds must comply with this standard to ensure that their processes meet the necessary quality and safety standards throughout the entire manufacturing cycle, from design to production.
- FDA Regulations: In the United States, the Food and Drug Administration (FDA) oversees medical device regulations. The FDA's regulations and guidance documents provide specific requirements for the design, manufacturing, and quality control of medical devices, including molds. Manufacturers must adhere to FDA regulations to obtain approvals or clearances for the medical devices produced using the molds.
- CE Marking: In the European Union, medical devices must bear the CE marking to demonstrate compliance with the relevant regulations. Molds used in the production of medical devices intended for the EU market must meet the requirements of the Medical Device Regulation (MDR) or In-Vitro Diagnostic Regulation (IVDR), depending on the type of device.
- Design Controls: Regulatory standards emphasize the importance of design controls, ensuring that the design and development processes are systematic and well-documented. This includes risk assessment, verification, validation, and documentation of design changes. Molds used in medical device production must be designed, tested, and documented to meet these controls.
- Traceability: Regulations often require manufacturers to maintain traceability of components, including molds, throughout their lifecycle. This includes tracking the materials used, manufacturing processes, inspections, and maintenance. This is crucial for identifying the source of any defects or quality issues.
- Documentation and Records: Regulatory compliance requires comprehensive documentation and records of the mold design, manufacturing processes, quality control measures, and any changes made over time. These records demonstrate that the molds and the devices produced meet the necessary standards.
- Risk Management: Medical device regulations emphasize risk management. Manufacturers must conduct risk assessments to identify and mitigate potential hazards associated with the molds and the components they produce. Mitigation strategies should be integrated into the mold design and production processes.
- Post-Market Surveillance: Regulatory standards require manufacturers to monitor the performance of devices in the market and promptly address any issues. Similarly, molds used in device production should be monitored for wear, maintenance needs, and any impact on product quality.
In summary, regulations and standards significantly influence the design of medical plastic molds by ensuring the use of suitable materials, adherence to quality management systems, compliance with biocompatibility and safety requirements, and proper documentation of the design and manufacturing processes. Designers and manufacturers of medical molds must have a deep understanding of these regulations to produce molds that contribute to the production of safe and effective medical devices.

Image courtesy: https://www.fictiv.com/articles/the-standards-for-plastic-injection-molding-in-the-medical-industry
Designing a plastic mold for a medical device is a multifaceted process that involves a comprehensive understanding of engineering principles, material science, regulatory requirements, and manufacturing considerations. Here is a detailed exploration of the key considerations when designing a plastic mold for a medical device:
- Regulatory Compliance and Standards:
- Adherence to regulatory standards, such as ISO 13485, FDA regulations, and CE marking requirements, is paramount. Regulatory compliance ensures that the mold and the resulting medical device meet safety, quality, and performance standards.
- The mold design should align with the intended use, classification, and regulatory pathway of the medical device. A clear understanding of regulatory guidelines influences every aspect of the mold's design and fabrication.
- Biocompatibility and Material Selection:
- Biocompatible materials for the mold are essential to prevent contamination of the medical device. The choice of mold material should consider its potential interactions with the medical-grade plastics, chemicals, and sterilization methods involved in the molding process.
- Compatibility extends beyond physical interactions to encompass potential leaching of harmful substances into the molded parts. Robust material selection mitigates risks associated with patient safety and device performance.
- Mold Geometry and Part Design:
- The mold's geometry must precisely mirror the intended shape, features, and dimensions of the medical device component. Meticulous attention to detail ensures that the molded parts meet stringent specifications and function as intended.
- Complex geometries, undercuts, and intricate features must be faithfully replicated in the mold to achieve accurate and high-quality components.
- Mold Cooling and Heating:
- Proper cooling channels are strategically integrated into the mold design to ensure uniform temperature distribution during the molding process. Effective cooling minimizes cycle times, reduces the risk of warping, and enhances the consistency of molded parts.
- In some cases, mold heating might be required to maintain optimal material flow and prevent premature cooling, particularly when molding high-temperature plastics.
- Ejection Mechanism:
- Designing an efficient ejection system is crucial for consistent and damage-free removal of molded parts from the mold cavities. The ejection mechanism, often employing ejector pins, must be carefully planned to prevent cosmetic or structural defects.
- Ejection should be balanced to ensure that parts are released smoothly without distortion or stress.
- Venting and Air Traps:
- Proper venting is incorporated into the mold design to allow air to escape as the plastic material flows into the cavities. Inadequate venting can lead to issues like air traps, where air is trapped within the mold, resulting in voids or incomplete filling of parts.
- The strategic placement of vents and air traps is essential to achieve defect-free components.
- Draft Angles and Surface Finish:
- The inclusion of draft angles in the mold design eases the ejection process and minimizes friction during part removal. Appropriate draft angles prevent damage to the part and mold, ensuring smooth operation.
- Careful consideration of surface finish in mold cavities influences the texture and appearance of the molded parts. The desired surface finish is designed to meet the specific requirements of the medical device.
- Gate Design and Location:
- Gate design and placement are critical to the overall part quality. The gate, where the plastic enters the mold, must be strategically positioned to minimize cosmetic defects, such as gate marks, and structural issues, such as weak points or weld lines.
- The gate type (e.g., edge gate, pin gate, hot runner gate) is selected based on material properties, part design, and aesthetic considerations.
- Injection Molding Parameters:
- The mold design should accommodate the optimal injection molding parameters for the chosen plastic material. Parameters such as injection pressure, temperature, and cooling time are meticulously calibrated to achieve consistent part quality and minimize defects.
- Parting Line and Mold Alignment:
- The parting line, where the mold separates into two halves, must be carefully determined to avoid flash or cosmetic imperfections. Precise mold alignment ensures uniform parting and prevents inaccuracies or defects at the parting line.
- Proper alignment contributes to the seamless assembly of multi-component medical devices.
- Tooling and Maintenance Considerations:
- Ease of maintenance is integrated into the mold design. Features that facilitate mold disassembly, cleaning, replacement of components, and routine maintenance extend the mold's lifespan and ensure the sustained quality of molded parts.
- Durable, wear-resistant materials are chosen to withstand the demands of production cycles while minimizing the need for frequent replacements.
- Quality Control Measures and Inspection Points:
- Mold design incorporates features that allow for quality control checks during production. Dedicated areas for dimensional measurement, visual inspection, and other testing methods ensure that each molded part meets specifications.
- Inspection points aid in identifying and addressing deviations in real-time, contributing to the consistency and reliability of the medical device components.
- Risk Management and Validation:
- Rigorous risk assessments are conducted during the mold design phase to identify potential hazards and mitigate associated risks. Validation activities, such as simulations, prototyping, and testing, validate the mold's performance and its impact on the medical device.
- The mold design is systematically refined to reduce potential failures, ensuring patient safety and regulatory compliance.
- Documentation and Traceability:
- Thorough documentation of the mold design process is essential for accountability and traceability. Detailed records encompass design iterations, simulations, validation results, changes, and maintenance activities.
- Documentation supports post-market surveillance, facilitates recalls if necessary, and enables continuous improvement of the mold design and production process.
- Post-Market Surveillance and Continuous Improvement:
- The mold design anticipates the need for post-market surveillance, enabling the monitoring of mold performance and its impact on device quality. Data collected during the device's lifecycle informs adjustments, updates, and enhancements to the mold design.
In essence, designing a plastic mold for a medical device demands meticulous attention to detail, collaboration between engineering and regulatory teams, and a deep understanding of both molding technology and medical device requirements. A well-designed mold is a cornerstone of producing safe, effective, and high-quality medical devices that meet the stringent standards of the healthcare industry.

Image courtesy: https://www.grainger.com/know-how/equipment-information/kh-how-does-autoclave-sterilization-work
The sterilization process has a significant impact on the choice of material for a medical plastic mold. Medical devices, including those produced using molds, must undergo sterilization to ensure they are free from pathogens and safe for use in clinical settings. The choice of mold material needs to consider how it will react to different sterilization methods while maintaining its structural integrity and functionality. Here's how the sterilization process affects the choice of material for a medical plastic mold:
- Sterilization Methods:
- Autoclaving: Autoclaving involves exposing devices to high-pressure steam at elevated temperatures. The repeated heating and cooling cycles can put stress on mold materials. Materials that can withstand repeated thermal cycling and maintain their mechanical properties are preferred.
- Ethylene Oxide (EtO) Gas Sterilization: EtO gas sterilization is a low-temperature method that can penetrate packaging and reach complex geometries. Mold materials must be compatible with EtO exposure and not undergo significant degradation or changes in mechanical properties.
- Gamma Irradiation: Gamma irradiation uses high-energy radiation to kill microorganisms. It's effective but can lead to polymer degradation over time. Mold materials should be radiation-resistant to maintain their structural integrity and dimensional stability.
- Chemical Sterilization: Chemical methods like hydrogen peroxide vapor or peracetic acid vapor sterilization are used for heat-sensitive devices. Mold materials should resist chemical interactions with sterilizing agents and not undergo degradation or changes in properties.
- Material Compatibility:
- The chosen mold material must be compatible with the specific sterilization method used for the medical device. Materials that can withstand the sterilization process without significant changes in mechanical properties, dimensional stability, or surface finish are essential.
- Material Properties:
- Sterilization processes can expose mold materials to extreme temperatures, humidity, chemicals, and radiation. The material's resistance to these conditions is critical. Heat resistance, chemical resistance, and radiation resistance are properties to consider.
- Mechanical Integrity:
- Sterilization processes should not compromise the structural integrity of the mold material. The material should maintain its strength, durability, and other mechanical properties after exposure to sterilization conditions.
- Dimensional Stability:
- Some sterilization methods involve temperature fluctuations, which can lead to thermal expansion and contraction. The mold material should exhibit good dimensional stability to prevent warping, distortion, or changes in mold dimensions.
- Surface Finish and Texture:
- The surface finish of the mold can affect the release of the molded parts. Sterilization methods should not deteriorate the surface finish or introduce contaminants that could affect the quality of the molded medical device components.
- Long-Term Performance:
- Mold materials should be chosen based on their ability to withstand repeated sterilization cycles over the mold's lifespan. Materials that maintain their properties and performance after multiple sterilizations contribute to consistent and reliable device production.
- Regulatory Considerations:
- Regulatory standards may require validation of the compatibility between mold materials and chosen sterilization methods. Proof that the mold materials do not negatively impact the quality or safety of the medical devices is crucial for regulatory compliance.
In summary, the choice of mold material for a medical device is influenced by its compatibility with various sterilization methods. The material's ability to maintain its properties and structural integrity after exposure to sterilization conditions is essential for producing safe and reliable medical devices. Proper material selection ensures that the mold remains a robust and consistent tool throughout the medical device's lifecycle.

Image courtesy: https://www.xometry.com/resources/injection-molding/injection-molding-safety-precautions/
Ensuring the quality and safety of medical plastic molds is an intricate and multifaceted process that requires a comprehensive approach across all stages of design, fabrication, validation, production, and maintenance. Molds are not only instrumental in shaping the medical device components but also directly influence the final product's performance, accuracy, and safety. Here's how the quality and safety of medical plastic molds are ensured:
- Robust Design Controls and Risk Management:
- The mold design process is governed by strict design controls. Detailed design input requirements are established to ensure that the mold design aligns with the intended use and performance of the medical device.
- Rigorous risk assessments are conducted to identify potential hazards, failure modes, and risks associated with the mold's design, fabrication, and use. Mitigation strategies are developed to address these risks and ensure patient safety.
- Thorough Material Selection and Compatibility Assessment:
- Mold materials are chosen based on their compatibility with medical-grade plastics, sterilization methods, and the device's intended use environment. Biocompatibility, chemical resistance, and sterilization resilience are meticulously considered.
- Compatibility assessments evaluate how the chosen materials will interact with the medical plastics, sterilization agents, and any chemicals used during production.
- Precise and Detailed Engineering:
- The mold design goes beyond aesthetics; it meticulously replicates the intended shape, features, and dimensions of medical device components. Gate locations, cooling channels, ejection mechanisms, and venting are engineered to prevent defects, ensure uniform part quality, and maintain dimensional accuracy.
- Validation Through Simulation and Prototyping:
- Computer-aided simulations and prototyping validate the mold design. Simulations predict how the mold will behave during production, helping identify potential issues like uneven cooling or defects.
- Prototyping allows for real-world testing of the mold's performance, ensuring that it can produce parts that meet specifications and withstand the stresses of molding.
- Adherence to Regulatory Standards:
- Regulatory compliance is non-negotiable. Molds, like medical devices, must meet relevant standards such as ISO 13485, FDA regulations, and CE marking requirements. This ensures that molds contribute to the production of devices that meet the highest safety and quality standards.
- Comprehensive Documentation and Traceability:
- Every detail of the mold's journey is meticulously documented. From initial design iterations and material selection to maintenance records and changes, traceability ensures accountability and the ability to track the mold's history.
- Stringent Manufacturing Quality Control:
- During mold fabrication, strict quality control measures are implemented. Each component of the mold undergoes inspections to verify its dimensions, surface finish, and functionality. Quality control ensures that molds are precision-engineered.
- Ongoing Post-Market Surveillance:
- Just as medical devices are subject to post-market surveillance, molds are monitored for performance, wear, and maintenance needs. Continuous monitoring ensures molds consistently produce high-quality components.
- Expertise and Training:
- Mold designers, engineers, and technicians are highly trained and experienced in the nuances of medical mold design and fabrication. Their expertise ensures that molds are designed, operated, and maintained to the highest standards.
- Strategies for Maintenance and Cleaning:
- Comprehensive maintenance and cleaning protocols are established and followed. Proper care not only prolongs mold life but also sustains the quality of the components they produce.
- Iterative Continuous Improvement:
- Data from post-market surveillance, production cycles, and quality control processes drive continuous improvement efforts. Any deviations or issues are thoroughly investigated, addressed, and used to refine mold designs and manufacturing processes.
- Supplier Quality Assurance:
- Suppliers providing mold materials and components are audited to ensure their quality and adherence to regulatory standards. Collaborating with reputable suppliers is paramount to upholding mold quality.
- Thorough Testing and Inspection:
- Rigorous testing and inspection protocols are established for the molded components. Random and systematic checks are conducted to ensure that molds consistently produce parts that adhere to specifications and quality standards.
In conclusion, the quality and safety of medical plastic molds are a culmination of meticulous attention to detail, regulatory adherence, validation, ongoing surveillance, and a commitment to continuous improvement. These molds are not merely tools for production; they play a vital role in upholding the highest standards of quality and safety in the medical device industry. By adhering to these principles, manufacturers ensure that molds contribute to the consistent production of medical devices that meet or exceed regulatory requirements and patient expectations.

Image courtesy: https://www.fictiv.com/articles/pros-cons-of-plastic-injection-molding-for-medical-products
Designing and manufacturing plastic molds for the medical industry is a highly complex and exacting process, driven by the need for precision, safety, and adherence to stringent regulatory requirements. The challenges encompass various technical, regulatory, operational, and industry-specific aspects that demand careful consideration and expertise. Here's are some of the challenges associated with designing and manufacturing plastic molds for the medical industry:
- Regulatory Compliance and Documentation:
- The medical industry is governed by strict regulatory standards to ensure patient safety. Designing and manufacturing molds that comply with standards such as ISO 13485, FDA regulations, and CE marking requirements necessitates meticulous documentation, validation, and quality control processes.
- Biocompatibility and Material Selection Challenges:
- Molds need to be fabricated from materials that are not only mechanically suitable but also biocompatible and chemically inert. Balancing these requirements while maintaining mold fabrication feasibility can be challenging due to limited material options that meet both criteria.
- Complex Geometries and Design Precision:
- Medical devices often have intricate and complex geometries to fulfill specific functions. Translating these complex shapes into mold designs that accurately reproduce them in molded parts while ensuring consistency, structural integrity, and dimensional accuracy requires advanced engineering and design expertise.
- Stringent Tolerances and Precision Manufacturing:
- Many medical devices demand exceptionally tight tolerances for proper functionality and compatibility with other components. Designing molds that consistently produce parts within these narrow tolerances necessitates precision manufacturing techniques and stringent quality control measures.
- Cleanliness and Contamination Control:
- Medical devices must be free from contaminants that could compromise patient safety. Ensuring that molds are manufactured in a controlled and clean environment to prevent potential contamination and maintain the quality of the final parts is a challenge that requires strict adherence to cleanroom protocols.
- Compatibility with Sterilization Methods:
- Molds must withstand the stress of various sterilization methods without degrading, warping, or introducing contaminants that could impact part quality. Accommodating different sterilization techniques while maintaining mold integrity is a complex endeavor.
- Multi-Material and Multi-Cavity Challenges:
- Certain medical devices require molds capable of handling multiple materials or featuring multiple cavities for efficient production. Designing molds that accommodate these complexities while maintaining precision, consistency, and part quality can be intricate.
- Validation and Verification Complexity:
- The validation and verification process for medical molds is extensive to ensure consistent part production that meets specifications and regulatory standards. Comprehensive validation requires time, resources, and thorough testing to ensure mold performance.
- Effective Risk Management:
- Rigorous risk assessments are essential to identify and mitigate potential hazards associated with mold design, manufacturing, and usage. Ensuring that these risks are appropriately addressed and minimized requires a deep understanding of both engineering and medical device regulations.
- Adapting to Post-Market Changes:
- The dynamic nature of the medical industry often leads to changes in device design, materials, or regulatory requirements after the launch of a product. Ensuring that molds can be adapted or modified without compromising regulatory compliance or device quality poses ongoing challenges.
- Balancing Cost and Quality:
- Achieving a balance between producing high-quality molds and managing costs can be challenging. The use of advanced materials and manufacturing processes required for high-quality molds may impact the overall production cost of medical devices.
- Specialized Expertise and Workforce:
- Successfully designing and manufacturing molds for the medical industry demands a workforce with specialized expertise in mold design, materials science, regulatory compliance, and precision manufacturing. Finding and training skilled professionals who understand both engineering principles and medical requirements can be challenging.
- Supplier Audits and Material Traceability:
- Ensuring the quality of materials used in molds requires audits of material suppliers and meticulous traceability to meet regulatory requirements. This involves establishing a robust supply chain management process.
In summary, designing and manufacturing plastic molds for the medical industry is a multifaceted endeavor that requires a comprehensive understanding of engineering principles, medical device regulations, materials science, and manufacturing techniques. Overcoming these challenges demands collaboration between skilled professionals across multiple disciplines to ensure the production of safe, effective, and high-quality medical devices.

Image courtesy: https://www.joharidigital.com/medical-device-prototype-development/
Rapid prototyping is a transformative tool that holds significant value within the design process for medical plastic molds. This technology-driven approach offers numerous advantages that revolutionize how molds are conceptualized, tested, refined, and brought to fruition. Rapid prototyping empowers designers and engineers to expedite development cycles, enhance precision, and streamline collaboration, ultimately contributing to the production of safer and more effective medical devices. Here's how rapid prototyping fits into the design process for medical plastic molds:
- Concept Visualization and Iteration Enhancement:
- Rapid prototyping bridges the gap between digital designs and tangible physical models. This visual representation aids stakeholders in comprehending the intricate mold structure, venting systems, gating locations, and other features that might otherwise remain abstract in the digital realm.
- Iterative design cycles are empowered by rapid prototyping. Designers can swiftly translate feedback into tangible modifications, accelerating the evolution of mold designs towards optimal outcomes.
- Reduced Time-to-Prototype and Accelerated Design Validation:
- Conventional mold fabrication methods are often time-consuming and resource-intensive. Rapid prototyping techniques, such as 3D printing, dramatically reduce the time required to materialize mold prototypes, facilitating quick and comprehensive design validation.
- Shortened development cycles allow designers to evaluate multiple design iterations within a condensed timeframe, enabling them to swiftly iterate on improvements and innovations.
- Validation and Testing Augmentation:
- Physical prototypes produced through rapid prototyping facilitate practical testing of mold designs. These tangible models can be utilized to evaluate fit, form, functionality, and ergonomics using actual medical device components.
- Incorporating medical-grade plastics into prototype molds enables simulation of the injection molding process. Designers can assess material flow, cooling efficiency, and part ejection dynamics, identifying potential enhancements or issues.
- Early Detection of Design Flaws:
- Rapid prototyping exposes design flaws in their infancy. Physical prototypes are examined for structural weaknesses, undercuts, interferences, and other geometry-related anomalies that might impede effective molding.
- The identification of these flaws at an early stage prevents their proliferation into full-scale production, mitigating costly rework and ensuring mold designs align with intended specifications.
- Facilitated Collaboration and Communication:
- The tangibility of physical prototypes fosters efficient communication across interdisciplinary teams. Designers, engineers, medical professionals, and stakeholders can physically interact with prototypes, promoting clear and concise collaboration.
- Enhanced communication minimizes misunderstandings and discrepancies, fostering a shared understanding of design intent and requirements.
- Cost-Efficient Iterations and Error Reduction:
- Unveiling design issues during the rapid prototyping phase curtails the risk of expensive errors during full-scale mold fabrication. Modifications are rapidly executed during prototyping, reducing the potential for costly revisions in later stages.
- Validation for Stakeholders and User-Centric Design:
- Involving clinicians and end-users is pivotal in medical device design. Rapid prototyping facilitates the creation of tangible prototypes that stakeholders can manipulate, leading to valuable insights that guide design adjustments for improved user experience.
- Functional Component Testing:
- Rapid prototyping permits the isolated testing of individual mold components, such as ejector pins, cooling systems, and gating structures. This approach validates their functionality and compatibility before integrating them into the complete mold assembly.
- Design Optimization and Innovation Nurturing:
- By physically assessing rapid prototypes, designers identify opportunities for optimization. Cooling channel layouts, gate positions, and other critical features can be refined to maximize efficiency, part quality, and production throughput.
- Support for Documentation and Regulatory Compliance:
- Tangible prototypes generated through rapid prototyping can be included in documentation for regulatory submissions. These prototypes visually demonstrate design concepts, validations, and compliance with regulatory standards.
- Feasibility Exploration for Novel Designs:
- Rapid prototyping acts as a feasibility assessment platform for inventive mold designs involving complex geometries or unconventional cooling strategies. This evaluation process enables designers to ascertain the viability of innovative concepts before committing to full-scale production.
In conclusion, rapid prototyping is an indispensable tool that seamlessly integrates into the medical plastic mold design process. Its influence extends beyond visualization to encompass accelerated validation, stakeholder engagement, error reduction, and design optimization. By embracing rapid prototyping, designers can navigate the complexities of mold design with greater precision, confidence, and efficiency, ultimately contributing to the development of medical molds that uphold the highest standards of safety, quality, and innovation.

Image courtesy: https://www.sciencephoto.com/media/270228/view/disposable-medical-instruments
Plastic molds are indispensable tools in the production of disposable medical devices, playing a pivotal role in shaping the landscape of modern healthcare. Disposable medical devices are designed for single-use purposes to minimize the risk of cross-contamination, enhance patient safety, and simplify clinical procedures. The integration of plastic molds into the manufacturing process ensures that these devices are produced with precision, consistency, and adherence to stringent regulatory standards. Here's are how plastic molds are utilized in the production of disposable medical devices:
- Conceptualization and Design Iteration:
- The journey begins with the conceptualization and design of the disposable medical device. Engineers, designers, and medical professionals collaborate to create 3D models that consider the intended clinical application, ergonomic considerations, regulatory compliance, and user experience.
- Mold Design and Fabrication Excellence:
- Once the device design is finalized, mold design takes center stage. Mold designers intricately craft plans for the mold's structure, cavities, gating systems, cooling channels, ejection mechanisms, and other critical features.
- The mold's design is meticulously engineered to replicate the precise geometry of the disposable medical device. Variables like material shrinkage during cooling are meticulously calculated to ensure that the molded parts align with the design specifications.
- Artistry of Mold Fabrication and Manufacturing:
- The virtual mold design transforms into a tangible mold through advanced manufacturing techniques. Precision machining, EDM (electrical discharge machining), CNC (computer numerical control) milling, and other state-of-the-art processes are harnessed to craft mold components with unparalleled precision.
- The mold's material is chosen carefully, taking into account biocompatibility, chemical resistance, and the compatibility with the chosen medical-grade plastic that the disposable device will be made from.
- Injection Molding Mastery:
- The chosen medical-grade plastic resin, often PVC, polyethylene, or polypropylene, is subjected to a controlled heating process before being injected into the mold under precisely controlled pressure and temperature conditions.
- The intricate cavities and channels within the mold outline the definitive shape of the disposable medical device. The injection molding process ensures uniform filling of the mold and subsequently cools the plastic to solidify, resulting in the final device form.
- Cooling and Ejection Precision:
- Cooling channels intricately woven into the mold play a pivotal role in controlling the cooling rate of the injected plastic. This precision ensures that the device solidifies uniformly, mitigating deformities, warping, or structural flaws.
- The completion of the cooling process signals the activation of the mold's ejection mechanisms. These mechanisms, which might involve pins, plates, or other components, facilitate the seamless release of the molded device from the mold.
- Unyielding Commitment to Quality Control:
- The molded disposable medical devices undergo rigorous quality control procedures. These encompass dimensional assessments, meticulous visual inspections, exhaustive functional tests, and thorough evaluations to ensure that the devices adhere to specifications and meet stringent regulatory requisites.
- Packaging and Sterilization Strategies:
- Following the quality control stage, the disposable medical devices are meticulously packaged within a sterile environment. The choice of packaging materials is meticulous, ensuring that the devices remain pristine, sterile, and safe until their designated use.
- Some disposable medical devices undergo sterilization processes such as gamma irradiation, ethylene oxide gas, or other methodologies to guarantee their sterility, paving the way for distribution and use in clinical settings.
- Distribution, Application, and Patient Care:
- Post-sterilization, the disposable medical devices are ready for distribution to healthcare institutions. These devices are versatile, serving multifaceted roles ranging from medication administration to diagnostic testing, wound care, and beyond.
- Efficacious Disposal Practices:
- Post-use, the disposable medical devices are safely and hygienically disposed of, aligning with healthcare waste management protocols. Their single-use nature circumvents the risk of cross-contamination and infection transmission, bolstering patient safety.
In summary, plastic molds are at the epicenter of the production of disposable medical devices, orchestrating a symphony of precision, compliance, and innovation. Through meticulous mold design, adept fabrication techniques, injection molding mastery, stringent quality control, and strategic sterilization, plastic molds contribute to the creation of disposable medical devices that epitomize safety, effectiveness, and regulatory adherence. These devices, designed for a single-use journey, impact patient care across the healthcare continuum while embodying the principles of innovation and patient safety that underpin modern medical advancements.
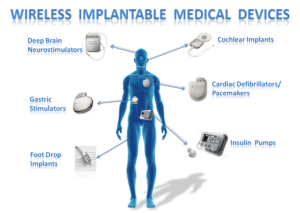
Image courtesy: https://groups.csail.mit.edu/netmit/IMDShield/
Plastic molds play a vital and intricate role in the production of implantable medical devices, serving as the linchpin that ensures these devices meet the highest standards of precision, quality, biocompatibility, and regulatory compliance. Implantable medical devices, ranging from orthopedic implants to cardiovascular stents, pacemakers, and intraocular lenses, have revolutionized modern healthcare by offering therapeutic, diagnostic, and supportive solutions that interact directly with the human body. The involvement of plastic molds in their production reflects the marriage of cutting-edge technology, materials science, engineering expertise, and healthcare innovation. Here's how plastic molds contribute to the production of implantable medical devices:
- Customized Design and Engineering Excellence:
- The journey begins with the conceptualization and engineering of implantable devices, often tailored to the specific needs and anatomical variations of individual patients. Designers and engineers collaborate closely to develop 3D models that encompass intricate geometries and functional considerations.
- The versatility of plastic molds facilitates the replication of these intricate designs, enabling the production of implantable devices that align perfectly with the intended anatomical or physiological requirements.
- Mold Design Precision and Fabrication Mastery:
- The foundation of the production process lies in the mold design, where meticulous plans are created to capture the complexity of the implantable device. These designs encompass cavity geometry, gating systems, cooling channels, and intricate features that are integral to the device's function.
- Mold fabrication is a testament to precision engineering, encompassing advanced manufacturing techniques such as CNC (computer numerical control) machining, EDM (electrical discharge machining), and intricate tooling. This results in mold components that mirror the intricacies of the design down to micron-level accuracy.
- Material Selection for Biocompatibility and Precision:
- The selection of mold materials is paramount, with a focus on biocompatibility and precision. Molds must be crafted from materials that are not only mechanically suitable but also biologically inert, ensuring that they do not introduce any harmful substances into the implantable devices.
- Injection Molding as a Craft of Excellence:
- Implantable devices often consist of medical-grade plastics that are compatible with the human body. During the injection molding process, these materials are heated and meticulously injected into the mold's cavities under precise pressure and temperature controls.
- The exquisite design of the mold, including gating systems, guarantees a controlled and uniform filling of the mold cavities, ensuring that the resulting implantable device faithfully represents the intended design.
- Mold Cooling and Solidification Prowess:
- Cooling channels intricately woven into the mold structure govern the cooling rate of the injected plastic. This precision ensures that the plastic material solidifies uniformly, minimizing the risk of defects, deformities, or inconsistencies.
- Ejection Mechanisms and Unwavering Quality Control:
- The intricate ejection mechanisms within the mold are orchestrated to release the solidified implantable device smoothly. Quality control becomes paramount at this stage, with meticulous dimensional checks, visual inspections, and functional assessments carried out to ensure that the devices meet design specifications and regulatory standards.
- Surface Finish and Biocompatibility Optimization:
- The surface finish of implantable devices assumes critical importance. The choice of high-quality molds and meticulous production techniques contributes to achieving the desired surface texture, which is essential for preventing bacterial adhesion and promoting biocompatibility within the body.
- Sterilization Compatibility and Material Resilience:
- Implantable devices must endure sterilization processes without compromise. The mold materials, as well as the design's resilience to various sterilization methods, are meticulously evaluated to ensure that the devices remain sterile and structurally intact.
- Regulatory Compliance and Documentation Precision:
- Regulatory compliance forms the bedrock of implantable device production. The molds utilized adhere to stringent regulatory standards, thereby ensuring that the devices themselves meet the safety, quality, and performance benchmarks set by regulatory authorities.
- Patient-Specific Manufacturing Advancements:
- Some implantable devices, such as customized orthopedic implants or dental prosthetics, require molds that are tailored to individual patient anatomies. These molds are painstakingly designed to accommodate patient-specific variations, leading to devices that offer optimal fit, function, and patient satisfaction.
- Prototyping and Iteration as Keys to Perfection:
- The rapid prototyping of implantable devices and their molds enables testing, validation, and iteration before committing to full-scale production. This iterative approach refines designs, hones manufacturing processes, and fine-tunes device characteristics, ensuring that the final product meets the highest standards.
- Collaboration, Expertise, and Patient Safety:
- The production of implantable devices requires a harmonious collaboration between diverse stakeholders, including designers, engineers, medical professionals, and regulatory experts. The collective expertise of mold designers, materials scientists, and manufacturing specialists ensures that devices are produced to exacting standards, prioritizing patient safety and clinical efficacy.
In summation, plastic molds are indispensable in the production of implantable medical devices, serving as the conduits that transform innovation into tangible solutions for patients. The seamless integration of advanced mold design, precision manufacturing, biocompatible materials, and rigorous quality control culminates in the creation of implantable devices that offer unparalleled precision, therapeutic value, and patient well-being. These devices embody the convergence of medical science and technological prowess, shaping the landscape of modern healthcare through their role in restoring health, enhancing quality of life, and exemplifying the highest standards of safety and efficacy.

Source: https://youtu.be/v7ilFyaGuXU
Maintaining cleanliness and hygiene in the production of medical plastic molds is of paramount importance to ensure the safety, quality, and integrity of the final medical devices. Contaminants, particulates, and impurities can compromise the functionality and biocompatibility of molds, which can subsequently affect the quality of the molded medical devices. Rigorous cleanliness protocols and cleanroom practices are essential to prevent contamination and uphold the stringent regulatory standards that govern the medical industry. Here's how cleanliness and hygiene are meticulously maintained in the production of medical plastic molds:
- Cleanroom Environment:
- The production of medical plastic molds often takes place within controlled cleanroom environments. Cleanrooms are designed to minimize airborne particles, microbes, and other contaminants that could settle on molds or components during the manufacturing process.
- Cleanrooms are equipped with specialized HVAC systems, air filtration units, and air pressure differentials that maintain a controlled and clean atmosphere. These measures prevent external pollutants from entering the cleanroom and safeguard the molds and equipment.
- Personnel Hygiene and Attire:
- Strict protocols govern the attire and personal hygiene of personnel working in cleanroom environments. Workers wear specialized cleanroom garments, including coveralls, gloves, hairnets, masks, and shoe covers, to prevent the introduction of particles and contaminants.
- Regular handwashing, thorough gowning procedures, and adherence to hygiene practices are enforced to minimize the potential for human-generated contaminants.
- Regular Cleaning and Maintenance:
- Molds and manufacturing equipment are regularly cleaned and maintained to prevent the buildup of contaminants. Cleaning routines involve using non-particulate-shedding cleaning agents and techniques that ensure no residues are left behind.
- Components that come into contact with molds, such as injection molding machines and auxiliary equipment, are also subjected to thorough cleaning and maintenance to prevent the transfer of contaminants.
- Particulate Control:
- Airborne particulates are a major concern in cleanroom environments. High-efficiency particulate air (HEPA) filters are employed to trap particles as small as 0.3 micrometers, significantly reducing airborne contaminants and maintaining a controlled atmosphere.
- Material Handling Protocols:
- Raw materials used in mold fabrication, such as plastics and mold release agents, are carefully stored and handled within the cleanroom environment. These materials are sourced from reputable suppliers and undergo stringent quality checks.
- Materials are often brought into the cleanroom in specialized packaging to prevent external contamination.
- Tool and Equipment Cleaning:
- Mold-making tools, machinery, and equipment are meticulously cleaned before use to prevent the transfer of contaminants to the molds. Cleaning processes involve the use of approved solvents and cleaning agents that leave no residue.
- Isolation and Segregation:
- Processes that generate higher levels of particles, such as machining or cutting, may be isolated from other processes within the cleanroom to prevent the spread of contaminants. Segregation ensures that processes with different cleanliness requirements do not interfere with each other.
- Regular Monitoring and Testing:
- Cleanroom environments are regularly monitored to ensure they adhere to specific cleanliness and contamination control standards. Particle counters and microbial monitors are used to assess the quality of the air and surfaces within the cleanroom.
- Training and Compliance:
- Personnel working in cleanroom environments receive extensive training in cleanliness protocols, gowning procedures, and contamination prevention measures. Regular training sessions reinforce the importance of maintaining a clean and hygienic environment.
- Quality Control and Documentation:
- Quality control processes are in place to ensure that the molds meet the desired specifications and regulatory standards. Detailed documentation tracks the cleaning, maintenance, and quality control activities, providing a clear record of adherence to cleanliness protocols.
In summary, maintaining cleanliness and hygiene in the production of medical plastic molds is a multi-faceted effort that involves a combination of controlled environments, strict protocols, personnel training, equipment maintenance, and stringent quality control. These measures collectively contribute to the creation of molds that uphold the highest standards of cleanliness, ensuring the safety, quality, and performance of the medical devices produced using them.